Cat. No: AX-AP01012A Size: 100 ug
Protein L, cl-APC conjugated
Introduction:
Protein L was isolated from the surface of bacterial species Peptostreptococcus magnus and was found to bind Ig(IgG,IgM,IgA,IgE and IgD) through L chain interaction. The recombinant protein contains four immunoglobulin (Ig) binding domains (Bdomains) of the native protein. Recombinant Protein L Protein, fused with the polyhistidine tag at N-terminus and a single cysteine at C-terminus. DTT-reduced protein migrates as a 36 to 38 kDa polypeptide.
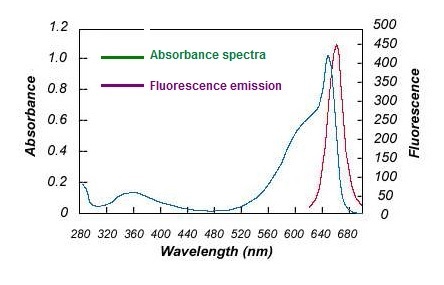
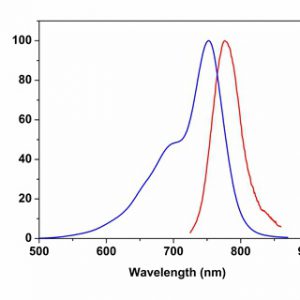
Allophycocyanin (APC), purified from Spirulina, is made up of alpha and beta subunits and is present as a trimer (αβ)3, which is unstable and susceptible to dissociation at low concentrations. The monomer, αβ, has a lower fluorescence quantum yield compared to the trimer and the maximal absorption is also shifted to 620 nm. The chemically cross-linked APC trimer (cl-APC) is much more stable than the native APC trimer, but still retains the same spectroscopic properties as the native APC trimer.
Protein L, cl-APC conjugated is a protein labeled by cl-APC.
Formulation: 100 µg of Protein L conjugated with cl-APC in PBS.
Excitation Laser: Red Laser (651 nm)
Application: In 0.1 ml PBS
Immunofluorescence (IF) 1: 50-1:200
Flow cytometry 1: 100-1:500.
The application notes include recommended starting dilutions; optimal dilutions/concentrations should be determined by the end user.
Specificity:
Despite this wide-ranging binding capability with respect to Ig classes, Protein L is not a universal immunoglobilin-binding protein. Binding of Protein L to immunoglobulins is restricted to those containing kappa light chains (i.e., k chain of the VL domain). Besides antibody, protein L is also suitable for binding of a wide range of antibody fragments such as Fabs, single-chain variable fragments (scFv), and domain antibodies.
Usage:
This product is for research use only. It is not approved for use in humans, animals, or in vitro diagnostic procdures.
| Categories |
|---|
Be the first to review “Protein L, cl-APC conjugate” Cancel reply
Related products
Flow Cytometry reagents & Conjugates
Flow Cytometry reagents & Conjugates
Flow Cytometry reagents & Conjugates
Flow Cytometry reagents & Conjugates
Flow Cytometry reagents & Conjugates
Flow Cytometry reagents & Conjugates
Cell-based immunoassay kits
Flow Cytometry reagents & Conjugates



Reviews
There are no reviews yet.